At the Laboratory for Optical Micromechanics and Imaging, directed by Dr. Seemantini Nadkarni, the major thrust of our research is the innovation and investigation of light-based technologies that interrogate the mechanical behavior of tissue, and on shedding light on the role of tissue mechanics in disease etiology, progression and prognosis. Our laboratory is located at the Wellman Center for Photomedicine at Massachusetts General Hospital, Harvard Medical School. The Wellman Center is the world’s largest academic research facility dedicated to the development of light-mediated, diagnostic and therapeutic technologies. The research efforts in our laboratory are directed towards both, fundamental and translational areas of Biomedical Optics to address major questions related to clinical medicine and biological sciences. Currently, our research activities are focused on five major areas of investigation
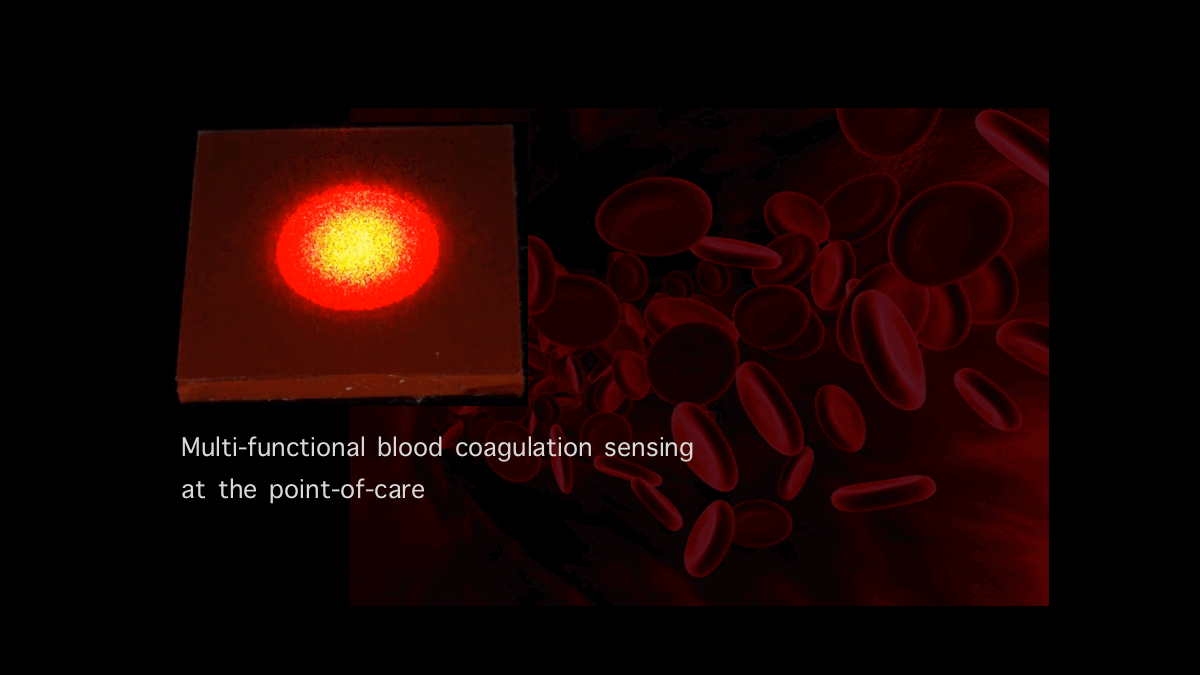
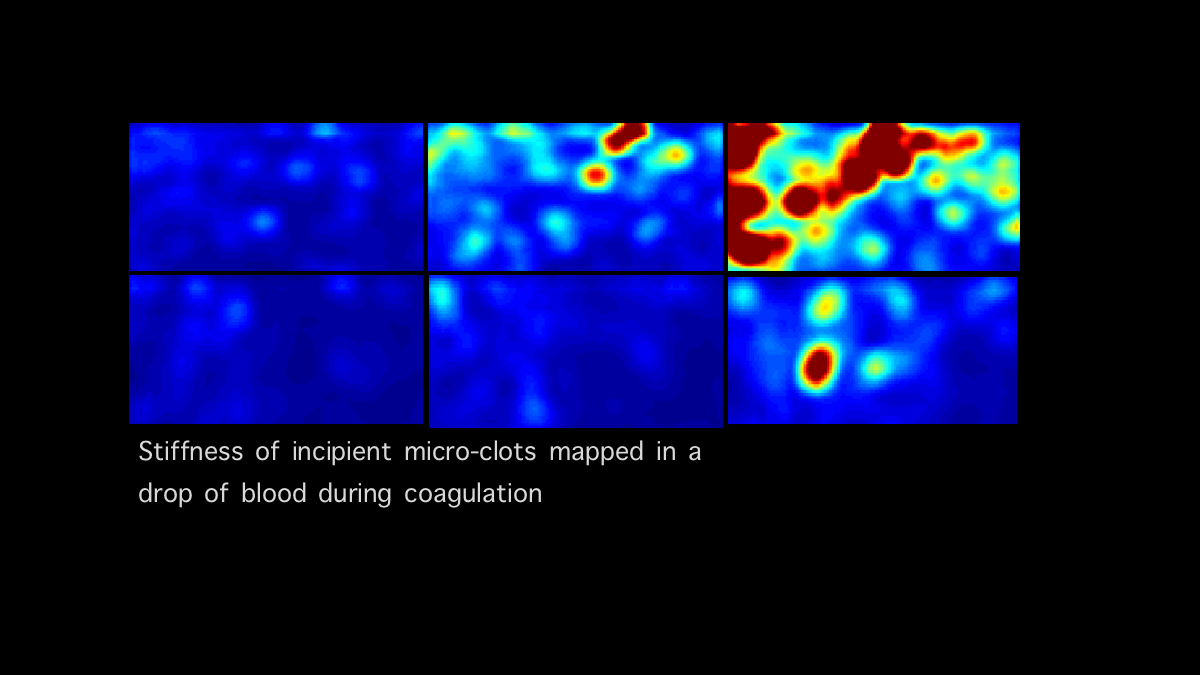
Blood Coagulation Sensing and In vitro diagnostics: Our team is developing new optical sensors that can quantify and report blood coagulation and platelet function profiles of patients within minutes using a drop of blood. These innovative technologies that can identify the cause of coagulation and platelet abnormalities at the patient’s bedside, are expected to have far-reaching clinical impact by rapidly identifying patients with impaired blood coagulation states so that they may be treated prior to the onset of life-threatening hemorrhage and acute thrombosis. In addition, these innovations will enable rapid coagulation testing in the home or primary care setting for over 15 million patients worldwide who routinely receive oral anticoagulants to prevent venous and arterial thrombosis, the world’s number one killer.
Polarization-sensitive optical frequency domain imaging (PS-OFDI): Intracoronary optical coherence tomography (OCT) and its next-generation counterpart, OFDI, have opened the exciting opportunity to visualize coronary atherosclerosis with exquisite micro-structural detail. An additional source of tissue contrast is measured using polarization sensitive (PS-OFDI) that detects the polarization state of reflected light to evaluate birefringence, a material property that is elevated in proteins with an ordered structure, such as collagen. Collagen is an important macromolecule that imparts mechanical stability to coronary plaques and the depletion of coronary collagen increases the propensity of plaque rupture and the ensuing risk of myocardial infarction. We are working in close collaboration with the Bouma Laboratory and our clinical collaborators, to apply the intracoronary PSOCT technology to address critical questions on the link between collagen metabolism, plaque rupture and medical therapy in patients.
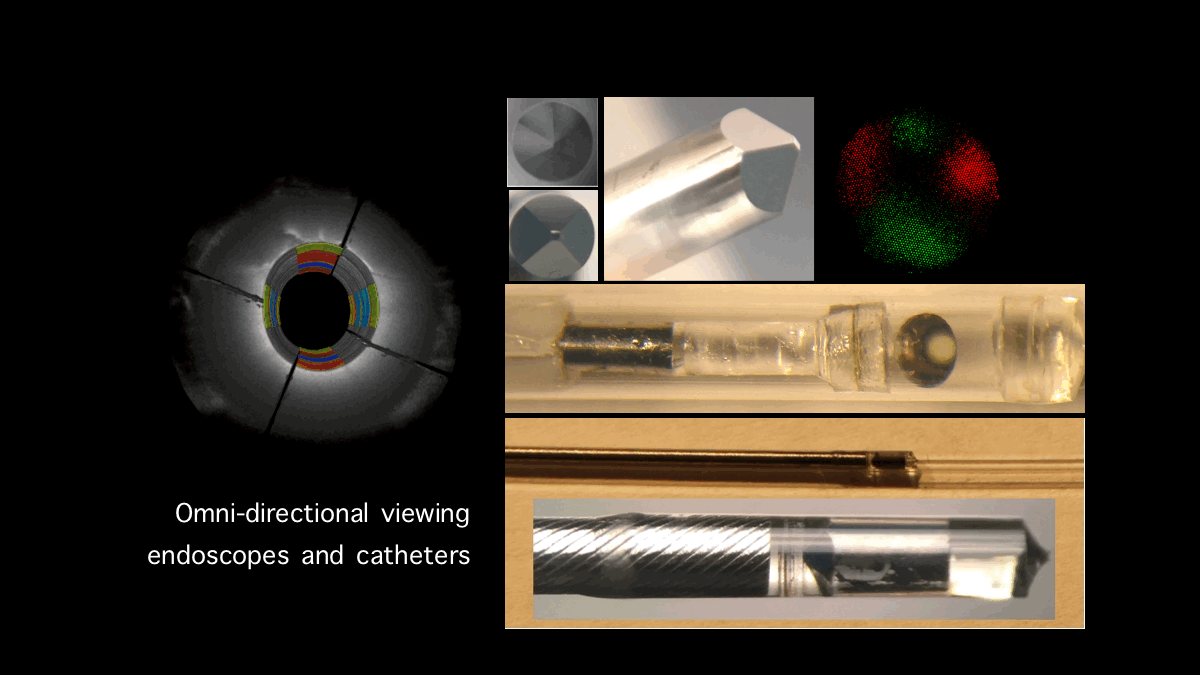
Intracoronary Plaque Biomechanics: Our laboratory has pioneered the development of a new technology, Intracoronary Laser Speckle Rheology (ILSR), for identifying mechanically unstable coronary plaques that cause myocardial infarction, the leading cause of death worldwide. We have conducted the first demonstration of ILSI in coronary and aortic vessels in vivo through the development of novel miniaturized, intraluminal catheters. This work has demonstrated for the first time that laser speckle patterns reflected from the coronary wall provide unique biomechanical signatures of plaque stability. We are currently working on incorporating the ILSR approach within a multi-modal imaging framework to identify critical mechanical and micro-structural features associated with myocardial infarction in patients.
Laser Speckle Microrheology (LSM): Our laboratory is leading the innovation of a new optical platform for light-based mechanical testing of tissue. This technology termed LSM, quantifies tissue stiffness with micro-scale resolution without perturbing, manipulating or contacting the sample. A number of micro-indentation devices and scanning probe microscopes are commercially-available to meet the need for micro-mechanical testing. These instruments however probe superficial depths over a small field of view, require several hours of measurement time, are costly and are unsuitable for in vivo assessment in living animals or patients. LSM is unique in that it probes depths up to several millimeters over a large field of view within minutes and can be operated with the push of a button. Moreover, since LSM does not contact or manipulate the sample during measurement, it offers several distinct advantages for operation in sterile environments where sample contamination by micro-indentation devices may pose a significant problem.
Extracellular Matrix Mechanics: Disease progression in tissues, from normal to pathological states, is often accompanied by changes in the intrinsic mechanical properties of the extracellular matrix (ECM). It is well recognized that virtually all cells are mechanosensitive, responding to mechanical cues from their microenvironment. Alterations in ECM mechanics can profoundly impact cell morphology, stiffness, migration, proliferation, differentiation and gene expression. Using light-based technologies including laser speckle microrheology and spectrally encoded confocal microscopy, our laboratory is working towards unlocking the complex liaisons between mechanical signaling evoked by the ECM and disease pathology in vivo.
We acknowledge all the funding agencies and entities that have supported our research efforts